 
For example, a quick glance at Figure AT5.2. That way, we can get a better look at the relationship. Often it is useful to plot data on a graph. As a result, fluorine is usually thought of as the most electronegative element. However, on many scales, fluorine would be the most electronegative atom here. As a result, noble gases are also given electronegativity values on this scale. The Allen scale just depends on the ability of an atom to interact with light, which is something even noble gases can do. Some electronegativity scales do not have values for the noble gases, because they are based on experimental measurements of compounds, and noble gases do not commonly form compounds with other elements. (Check the table!) Mendeleev said that the atomic weights must not have been determined correctly, but they were correct.\): The Allen electronegativity values of the second-row elements. For instance, tellurium (Te) was clearly a chalcogen, in the oxygen family, and iodine (I) was clearly a halogen, based on their properties, but the weights were wrong. Notice how the the valences repeat every 7 elements when they are arranged according to atomic mass.īut there were some problems with the table. Here's a smaller version of the periodic table that leaves out the elements Mendeleev and Meyer found most problematic (transition metals, rare earths) and the group that hadn't been discovered yet (noble gases). Mendeleev used this to predict the properties of undiscovered elements. The modern version of this periodic arrangement is the Periodic Table.Īlso, within a group (sharing a valence) properties like density, boiling point, heat capacity, etc follow a simple progression. Using Cannizzaro's atomic weights, Mendeleev and Lothar Meyer made a great discovery, the periodic law: If you arrange the elements by their atomic weights, there is a periodic repetition in properties such as valence. aluminum oxide, Al 2O 3: aluminum has a valence of 3īy the 1860s, ~60 elements were known.hydrogen sulfide, H 2S: sulfur has a valence of 2.lithium oxide, Li 2O: lithium has a valence of 1.methane, CH 4: carbon atoms have a valence of 4.Valence is the number of connections an atom tends to form. Scientists soon observed patterns in the valence of the different elements. As you go down the periodic table, it becomes easier to remove an electron from an atom (i.e., IE decreases) because the valence electron is farther away from the nucleus. Once Cannizzaro convinced most chemists to accept it, chemists were able to study the actual atomic weights and formulas. Avogadro's hypothesis let chemists figure out atomic weights and formulas together. 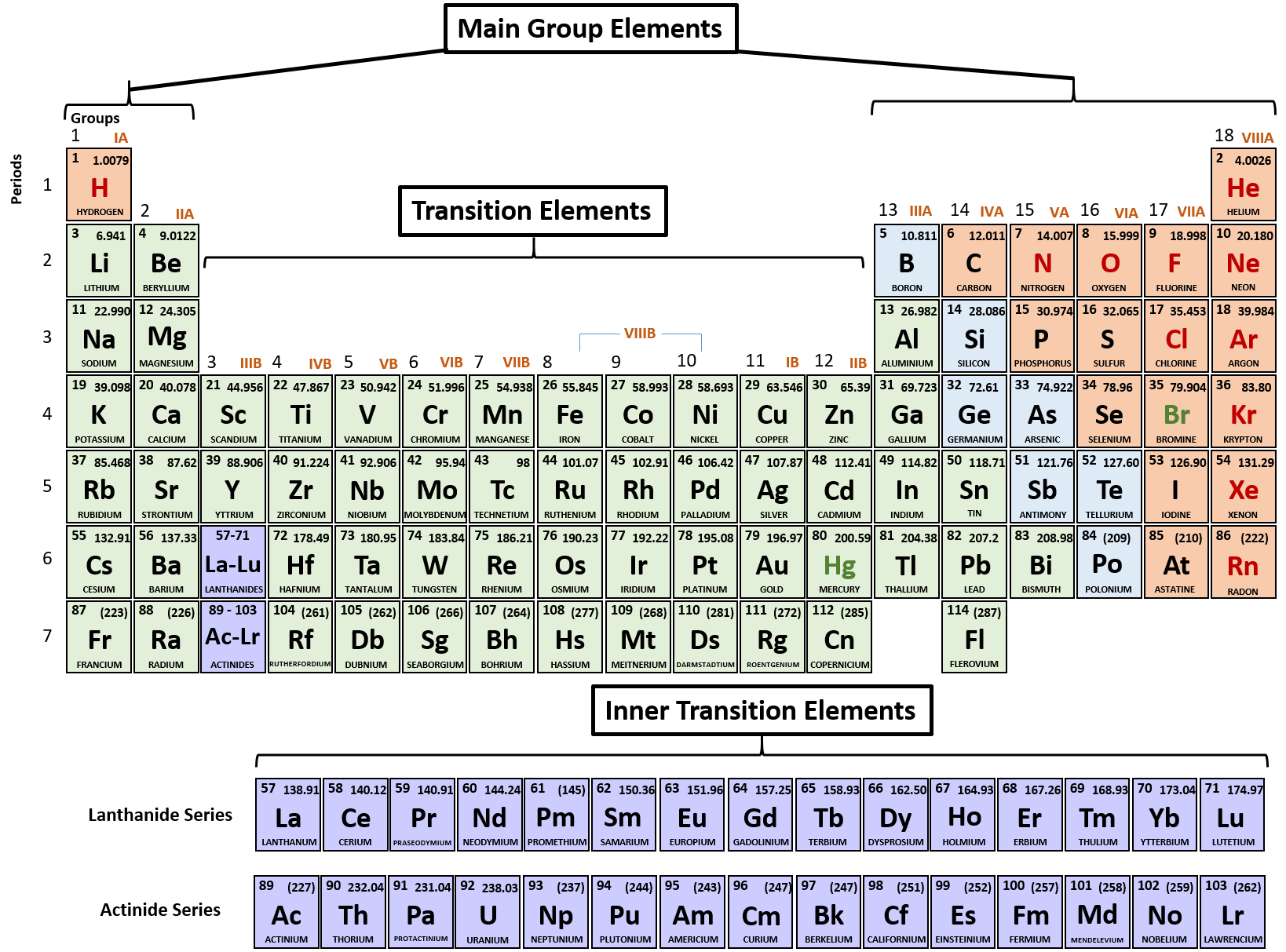
His paper explaining how to calculate molecular weights was distributed to everyone, including Julius Lothar Meyer, who wrote that when he read it "doubts disappeared and a feeling of quiet certainty took their place". 
Eventually, Cannizzaro revived Avogadro's hypothesis at a big meeting of chemists in 1860. However, Berzelius, who was working hardest on this problem, didn't believe either Avogadro or Faraday.īerzelius' beliefs held up the progress of science for about 50 years. The other was Faraday's law, which measured masses of elements produced by a set amount of current. One was Avogadro's hypothesis based on Gay-Lussac's law, which allowed chemists to relate the equivalent masses to equivalent volumes. There were two good ways available to figure this out. But what about elements? For instance, what mass of element A reacts with 1 g of element B? Knowing these numbers could be very useful, but it was hard to relate the equivalent masses to the actual masses of the atoms because they didn't know the formulas. For instance, what mass of acid A neutralizes base B? These numbers were easy to measure and practically useful. Many chemists were interested in knowing the "chemical equivalents" of different substances.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
